
In statistical mechanics the equation representing the third law of thermodynamics is as follows: Third Law of Thermodynamics - Mathematical Explanation The SI units representing entropy are J/K (joules/degrees Kelvin).An isothermal process states when the heat change (Q) is divided by the absolute temperature (T), it results in a change in entropy (ΔS).Chemical complexity results in higher entropy.Hard and brittle materials have lower entropy than pliable solids like metals.Dissolving gas in water lowers the entropy. 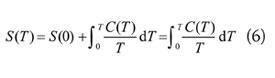
Mixing of solids and liquids results in higher entropy.Melting and evaporation of the system lead to higher entropy.The physical properties of entropy are as follows: The more the energy is dispersed, the greater would be the entropy.While work is done, the usable energy is converted to unusable energy.Nature plays a vital role in creating disorder compared to order, like ageing, rusting, decaying, etc.Any variation in entropy associated with a process from its initiation to its end is denoted by ΔS.The microstate wherein the system has the lowest energy is termed the system's ground state.This means the more microstates the closed system has, the higher its entropy will be. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |



 RSS Feed
RSS Feed
